Introduction
Since late 2020, there have been various COVID-19 vaccines that have been developed, approved and recommended in Canada based on research and epidemiological reports. News of these COVID-19 vaccines being developed and approved have been met with excitement and hope, but also some skepticism, particularly among older adults and some of the most vulnerable members of our society.
There are many legitimate questions and misconceptions out there about COVID-19 vaccines, how they came to be and what they mean for our personal health. It is important to address these concerns as Canada moves away from a crisis phase and progresses towards the long-term management of COVID-19.
Here are some common questions arising about COVID-19 vaccines among older Canadians along with evidence-informed answers to help keep you up to date with the facts.
How do vaccines work?
Our body may come across various bacteria, viruses or fungi that can cause diseases. These are known as pathogens. To fight off these disease-causing organisms, our body’s immune system develops antibodies that are produced based on a part of the pathogen called an antigen.
This helps create protection against the disease, which is known as immunity. In our body, we have thousands of different antibodies for specific pathogen-related antigens. When our body encounters a new pathogen, like the COVID-19 virus, it will take time to produce the specific antibodies. By the time our body is ready to fight back, the infection can already have caused a lot of damage that can sometimes lead to severe illness and death. But once it fights off that virus, our body also creates antibody-producing memory cells that helps it remember how to fight off that particular virus. Therefore, the next time you come into contact with the same virus, your body remembers it and is able to quickly assemble its defence system against it.
This is where vaccines can be advantageous. Vaccines contain weakened virus, inactive antigens or a blueprint to produce antigens that triggers an immune system response. They allow the body to ramp up an immune response against a variety of pathogens. This means that later, if your body comes across an actual pathogen you have been immunized against, it can quickly recognize and respond to it immediately, and fight it off before it causes serious health issues.
How do mRNA vaccines work and
which ones are available?
mRNA or messenger RNA, is a naturally occurring molecule in our bodies and gives instructions to our bodies to make necessary proteins, similar to a recipe card.
Prior to the COVID-19 pandemic, researchers had been studying and working with mRNA-based treatments for decades. As soon as the necessary information about the virus that causes COVID-19 was available, scientists began designing the first mRNA COVID-19 vaccines.
In these vaccines, synthetic mRNA is used to tell the body to make a harmless piece of the spike protein antigen that is found on the surface of the actual COVID-19 virus. These vaccines work by delivering mRNA instructions or a recipe card that directs the body to produce a small amount of the spike protein antigen. Our body recognizes that this antigen doesn’t belong and ramps up an immune response by producing protective antibodies against the COVID-19 virus’ spike protein antigen, so if our body later comes in contact with an actual COVID-19 causing virus, it immediately knows how to fight it off. The mRNA in the vaccine does not last long in the body as it quickly gets broken down. The mRNA from the vaccine cannot get into your DNA or alter it in any way.
mRNA vaccines are an exciting scientific advance and will allow us to design vaccines for other viruses as well, such as influenza, respiratory syncytial virus (RSV), Zika, rabies and cytomegalovirus (commonly known as CMV). mRNA vaccines are also being studied as a novel way to fight cancer.
There are various mRNA COVID-19 vaccines approved in Canada for adults, targeting different variants of the SARSCOV-2 virus. Over the past three years the following mRNA vaccines have been approved: ComirnatyÒ , Comirnaty® Original & Omicron BA.4/BA.5, Spikevax BivalentTM (Original/Omicron BA. 4/ BA.5) and Spikevax BivalentTM (Original/ Omicron BA.1).
In addition to these vaccines, two new mRNA vaccines have been approved this fall to target one of the more recent virus strains: Comirnaty® Omicron XBB.1.5 and Spikevax® XBB.1.5.
How do protein subunit vaccines
work and which ones are
available?
Protein subunit vaccines have been used for several decades for the protection against various diseases including pertussis and hepatitis B.
These vaccines contain purified proteins of the virus that are developed through a well-established process. First, a piece of the virus’s genetic material that holds instructions to develop the COVID-19 virus’ spike protein is inserted into another cell, allowing the production of the protein. The protein is then purified and inserted into the body to trigger an immune response. To enhance the immune response of the protein antigen, the vaccine also includes the adjuvant Matrix-M. Adjuvants have been used safely across various vaccines.
Protein subunit vaccines are not able to cause infection as it does not contain the virus.
Currently, the NuvaxovidTM COVID-19 vaccine is the only protein subunit COVID-19 vaccine available for adults in Canada.
What are the newest formulations
of COVID-19 vaccines?
Currently, there are newer Omicron strains that are more prevalent in Canada compared to what is being targeted through the bivalent mRNA COVID-19 vaccines. These new Omicron strains are able to better avoid antibodies from the body compared to previous Omicron strains.
In the fall of 2023, a new set of COVID-19 vaccines have become available in Canada that specifically target one of the more recent Omicron strains. These vaccines do not include the original strain of the SARSCoV-2 virus, and instead only comprise one of the newer Omicron strains. As a result, they are expected to provide a better immune response in comparison to the currently available COVID-19 vaccines, including the bivalent mRNA COVID-19 vaccines.
Also, it is estimated that thousands of hospitalizations and deaths in Canada can be prevented over the next year through an additional vaccine dose being offered this fall.
For this reason, NACI has recommended that individuals receive the XBB.1.5- containing COVID-19 vaccine, regardless of the number of vaccine doses they have previously received. The time interval for vaccination, depends on previous COVID-19 vaccine and/or infection history (please see page 15 for recommendations for older adults).
The Comirnaty® Omicron XBB.1.5 (Pfizer-BioNTech) and Spikevax® XBB.1.5 (Moderna) were approved for use by Health Canada in September 2023, with a new formulation of COVID-19 vaccine by Novavax expected to be approved by Health Canada soon. Already, preclinical and clinical data demonstrated that the new Moderna and Pfizer-BioNTech vaccines developed robust immune responses against current SARS-CoV-2 XBB* Omicron virus strains.
COVID-19 vaccines have been
developed very fast. Have important
steps been skipped in their
development?
According to the Health Canada’s website, all of Canada’s approved COVID-19 vaccines have:
- met the normal requirements for a vaccine’s approval, including all of the usual safety, quality and efficacy requirements for vaccines, and no requirements were overlooked in order to approve the use of these vaccines; and
- their quality, safety and effectiveness monitored on an ongoing basis.
Countries and companies across the world have banded together and collaborated in a way we haven’t seen in the past to help develop COVID-19 vaccines. Health agencies and vaccine researchers and manufacturers have prioritized the development of COVID-19 vaccines by investing huge sums of money, redeploying staff and developing several collaborations to work on COVID-19 vaccine-related efforts. None of this occurred at the expense of safety, and due diligence was absolutely done in the clinical trials that were required to demonstrate both their safety and efficacy.
Other factors that accelerated the creation of the approved COVID-19 vaccines include:
- The development of our earliest COVID-19 vaccines were based on decades of research that were conducted on other strains of coronavirus prior to COVID-19, such as Middle East Respiratory Syndrome (MERS) and SARS-CoV (SARS).
- Additional advances in science and technology made the development of new vaccines easier; once the virus was genetically sequenced, which occurred soon after the COVID-19 virus was discovered, scientists could quickly get to work to create a variety of vaccine candidates and start clinical trials.
- Strong international collaboration among scientists, health professionals, researchers, industry and governments, including ample funding to implement the large clinical trials needed to more quickly test and establish the safety and effectiveness of the vaccines being developed.
Should people take acetaminophen
(also known as Tylenol) or nonsteroidal anti-inflammatory
drugs before getting vaccinated
to prevent post-vaccination
symptoms?
Even though these medications may be taken to manage symptoms post-vaccination, it is not recommended that such medication be regularly used before or during vaccination. However, in case these medications have been taken, vaccination can still be administered.
If needed to manage symptoms post-vaccination, acetaminophen is preferred for use by older persons.
I already had COVID-19. Do I need
to get the vaccine?
Even if you have already contracted COVID-19 in the past, you will still benefit from getting vaccinated because the immunity one develops after having both an infection and vaccination (known as hybrid immunity), have led to low rates of future infections and severe disease. This is especially the case with individuals who have hybrid immunity with an infection from a more recent Omicron strain. It is recommended that for individuals who have not started or completed their primary series doses, they wait eight weeks from the positive test or symptom onset before getting vaccinated. For individuals who have completed their primary series, it is recommended they wait six months from the time of the previous infection to obtain their next vaccine dose.
There are no safety concerns with receiving vaccination after a recent infection, which has been found to be well tolerated; however, the reason for the time intervals is due to the understanding that an individual’s resulting immune response is higher when there is a longer time between an infection and vaccination.
Even if hybrid immunity was already obtained from prior vaccination and infection, an individual’s protection against infection eventually decreases over time. This is one of the reasons why doses are being recommended if people have not been vaccinated against or experienced a recent COVID-19 infection in the last six months.
Can I get the influenza vaccine
and COVID-19 vaccine at the same
time?
Not only the influenza vaccine, but COVID-19 vaccines may also be given at the same time, or any time before or after other vaccines (e.g., respiratory syncytial virus, pneumococcal and shingles).
Please speak with your health care provider regarding other vaccines that are recommended for you.
For more information on recommended vaccines for older adults, please see page 26.
I’m an older adult. Should I get
a COVID-19 vaccine?
Older adults have been found to be greatly impacted by COVID-19 infections, with adults 60 years and older making up 70 per cent of COVID-19 hospitalizations and 92 per cent of COVID-19 cases deceased in Canada.
The COVID-19 vaccines that have been approved in Canada for use in older Canadians have been found to be both extremely safe and effective.
There are many benefits associated with receiving the COVID-19 vaccine — they will lower your risk of getting sick if you are exposed to the virus and reduce risks of severe effects (e.g., hospitalizations, deaths). Receiving the COVID-19 vaccines will also protect those around you that may also be at risk of getting seriously ill. The Public Health Agency of Canada and Canada’s medical and nursing associations recommend that all older Canadians get vaccinated when they are able to do so.
Vaccines are especially important with the current variants of SARS-CoV-2 being better able to avoid antibodies from vaccines and previous infection. In the fall of 2023, new formulations of COVID-19 vaccine have become available that are targeting these variants and are expected to provide better protection against these variants compared to currently available vaccines.
Are the COVID-19 vaccines
effective and safe in older adults?
Yes, the Pfizer-BioNTech, Moderna and Novavax vaccine trials enrolled sizable numbers of older adults in their original vaccine trials to establish that their COVID-19 vaccines are both safe and effective.
While the overall efficacy of the Pfizer-BioNTech, Moderna and Novavax vaccines vary at preventing laboratoryconfirmed COVID-19 illness in people, their efficacy in preventing hospitalization and death has been found to be the highest for individuals fully vaccinated in the clinical trials.
The updated XBB.1.5-containing COVID-19 vaccines are expected to perform better against the current strains compared to previously available COVID-19 vaccines.
Already, preclinical and clinical data demonstrated that the new PfizerBioNTech and Moderna vaccines developed robust immune responses against current SARS-CoV-2 XBB* Omicron virus strains.
Comparing differing efficacy rates between vaccine trials is not recommended because each trial has slightly different efficacy endpoints — so these cannot be equally compared to each other. In addition, the clinical trials related to different COVID-19 vaccines were done in different countries at different times during the pandemic, and the amount of COVID-19 as well as the number and type of variants that were circulating during these trials could have impacted their efficacy results. What is important to remember is that all of the currently available vaccines are very effective in preventing severe illness, hospitalizations and COVID-19-related deaths.
However, regardless of vaccination, due to the uncertainties regarding COVID-19 and the length of protection from recommended vaccines and previous infection, we should not stop other important public health measures to prevent the spread of COVID-19. Mask wearing in public, physical distancing, frequent hand washing and avoiding crowded settings remain important strategies.
Which COVID-19 vaccines should
older adults take and how far
apart?
Older adults are recommended to receive the updated XBB.1.5- containing COVID-19 vaccines.
This vaccine is expected to provide a better immune response against the current circulating COVID-19 strains compared to the earlier COVID-19 vaccines, and will also improve protection against infection, symptomatic and severe disease that might have been reduced since a person’s last vaccination or infection. Currently, there are approved Moderna and Pfizer-BioNTech COVID-19 vaccines with the new XBB.1.5-containing formulation. Over the coming months it is expected the Novavax XBB.1.5- containing COVID-19 vaccine will also be approved for use in Canada. mRNA vaccines (Pfizer-BioNTech and Moderna) remain the preferred COVID-19 vaccines, with the Novavax vaccine recommended to be used for those who are unwilling or unable to receive the mRNA vaccines.
For individuals who have never received any doses of COVID-19 vaccines (have not started primary series doses) are recommended to receive one dose of the XBB.1.5-containing COVID-19 vaccine. If they had a recent COVID-19 infection, vaccination may be delayed by eight weeks.
For individuals who have started their primary series doses, but only received one dose of an earlier COVID-19 vaccine, are recommended to complete the series by receiving one dose of the XBB.1.5- containing vaccines. This dose can be received if it has been at least eight weeks since receiving the previous COVID-19 vaccination or infection.
For individuals who have completed their primary series doses (at least two doses of earlier COVID-19 vaccines) are recommended to receive an additional dose of the XBB.1.5-containing COVID-19 vaccine. This additional dose can be received if it has been at least six months since receiving the previous COVID-19 vaccination or infection.
NACI will provide further guidance on whether additional doses of the XBB.1.5-containing COVID-19 vaccine are recommended.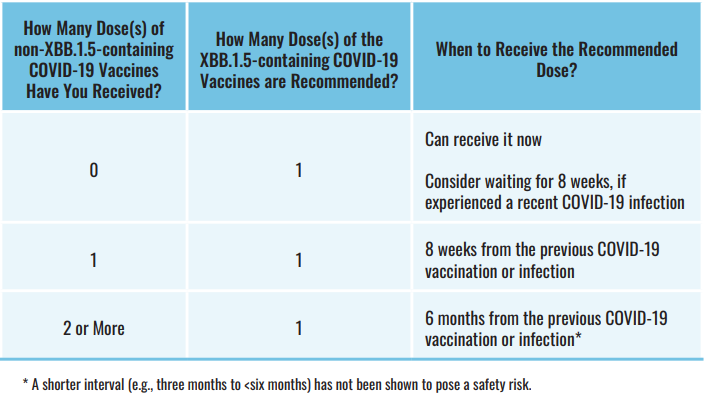
What are the side effects of
the COVID-19 vaccines in older
adults?
Vaccine side effects mean that the body’s immune system is kicking in and getting ready to recognize and fight off the virus if it ever encounters it in the future.
The side effects associated with COVID-19 vaccines are usually mild and no different in older adults than in the rest of the population.
What is most reassuring is that no clear pattern of serious or unexpected adverse events have occurred among the hundreds of millions of older adults who have been vaccinated against COVID-19 thus far.
Across the XBB.1.5-containing COVID-19 vaccines, the very common side effects may include pain where you were injected, tiredness, headache, muscle aches, chills and a fever. These side effects can last for a day or two, and rarely more than a few days.
Vaccine side effects are typically a sign that your immune system is doing exactly what it is supposed to do: working and building up your immunity to protect you from what you are being vaccinated against.
In fact, as older adults tend to have weaker immune systems than younger people, they appear to be developing side effects at a lower rate, but are still achieving the same level of immunity or protection that younger adults achieve with these vaccines.
You can have an allergic reaction to any medicine or food. It is possible that some people are allergic to an ingredient in the COVID-19 vaccines, but it is important to remember these allergic reactions are relatively rare. It is recommended that if you have allergies, including severe allergies that require you to carry an EpiPen, you should discuss the vaccine with your doctor who can assess your risk and provide more information about how to get vaccinated safely.
I am caring for an older adult that
does not want to get the vaccine.
How can I convince them that it is
safe?
You can show an older adult COVID-19 Q&As such as this one or any of the links below that answer questions specifically related to older adults.
Also emphasize to the older adult in your life that COVID-19 is a serious illness and that 92 per cent of Canada’s COVID-19 deaths have been among older Canadians 60 years of age and older.
It is understandable that older adults may be wary of the COVID-19 vaccine, but knowing the facts that any potential risks of side effects related to getting the vaccine are likely far better than the risk of dying from COVID-19 should provide them reassurance, especially when hundreds of millions of older people around the world have safely received the COVID-19 vaccine so far.
Should older adults with health
conditions get the COVID-19
vaccine?
Studies have shown that not only do adults with health conditions have a higher risk for severe outcomes from COVID-19, but the risk increases with the number of conditions.
For these reasons, vaccination is particularly important for the conditions listed below. If you have at least one of these conditions, please speak with your health care provider regarding vaccination this fall 2023.
These medical conditions include (as noted by Canada’s Public Health Agency of Canada):
- Cancer
- Cerebrovascular disease
- Chronic kidney disease
- Certain chronic liver diseases
- Certain chronic lung diseases
- Cystic fibrosis
- Diabetes mellitus, type 1 and type 2
- Disabilities
- Heart conditions
- HIV infection
- Certain mental health disorders
- Obesity
- Primary immunodeficiency diseases
- Smoking, current or former
- Solid organ or blood stem cell transplant
- Tuberculosis
- Use of corticosteroids or other immunosuppressive medication
Is it safe to get the COVID-19
vaccine in older adults with health
conditions?
Generally, it is safe for individuals with health conditions to get the COVID-19 vaccines.
However, it has been noted that those with either bleeding disorders or receiving blood thinner medications should inform their health care providers, in order to avoid the risk of bleeding, bruising or developing a haematoma (pooling of blood under the skin) following vaccination. It is also advised that adults who have any health condition or are taking medication notify their health care provider to avoid any side effects and ensure proper use of the vaccines.
Should someone with allergies get
the COVID-19 vaccine?
Individuals with allergies whether related or not to the COVID-19 vaccine should discuss with their health care provider about getting the COVID-19 vaccine. For individuals who are advised to take the COVID-19 vaccine, the length of time observed after vaccination may vary depending on the type of allergy (15 to at least 30 minutes).
Even for individuals who had a mild to moderate allergic reaction to a previous dose of COVID-19 vaccine are advised to discuss this further with their health care provider, as they may be able to be receive another dose.
Is it safe to get the COVID-19
vaccine in older adults who are
immunocompromised?
Immunocompromised patients, or those with weakened immune systems, tend to be at increased risk from becoming seriously ill and dying from COVID-19.
This can include older persons living with cancer, HIV, those who are transplant recipients, or are taking steroids or other medicines to treat certain medical conditions, called immunosuppressants, that lower the body’s ability to fight some infections.
Because of their increased risk of becoming seriously ill and dying from COVID-19 infections, immunocompromised people should receive the COVID-19 vaccines if there are no absolute contraindications. As none of the currently approved vaccines include live virus, there is no risk of getting infected with the actual virus when getting vaccinated.
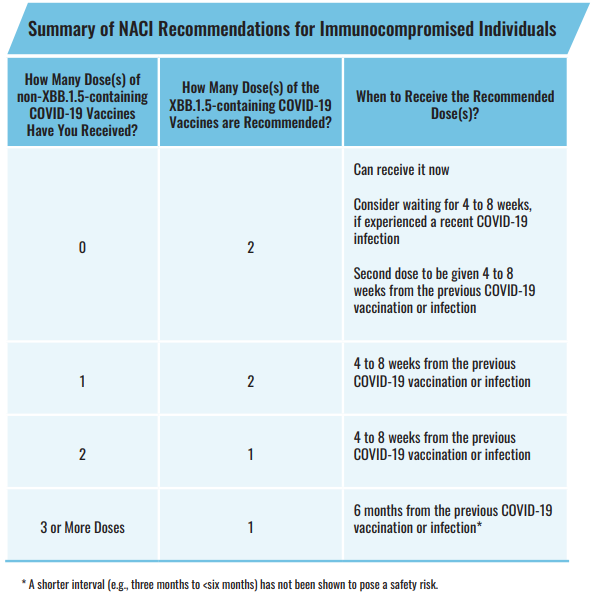
The need for COVID-19 vaccines by adults who are immunocompromised is clearly highlighted as they are recommended to receive two to three doses, four to eight weeks apart, for their primary series. Other adults are only recommended to receive one to two doses, eight weeks apart for their primary series. Please see table below for a summary of NACI recommendations for individuals who are immunocompromised.
Overall, for older Canadians with wellcontrolled immune disorders, medical experts emphasize that the positive benefits of the COVID-19 vaccines almost always outweigh any risks, and immunization is recommended.
Furthermore, it is always good to check for the latest information and advice around the safety and effectiveness of these vaccines with your doctor who knows your overall medical situation well.
Is it safe and recommended for
older persons living with dementia
to get the COVID-19 vaccine?
Age is the greatest risk factor for having dementia. People living with dementia often live with at least one other chronic condition and have been shown to be at much greater risk, compared to people without dementia, of getting infected, seriously ill and dying from COVID-19.
People living with dementia are more likely to have problems remembering and understanding recommended public health measures, such as physical distancing and wearing a mask in the presence of others, which significantly increases their risk of getting infected from COVID-19. This is why the Public Health Agency of Canada, Canada’s medical and nursing associations, and the Alzheimer Society of Canada all recommend that all older Canadians, including those living with dementia, get vaccinated when they are able to do so.
There is some concern that some of the limited side effects associated with COVID-19 vaccines, like a sore arm, headache, muscle aches, fatigue, fever or diarrhea that can last for a few days may cause some increased confusion in a person living with dementia, but these post-vaccination side effects can usually be well-managed with acetaminophen or other treatments.
Millions of older persons around the world living with dementia have now safely received their COVID-19 vaccines and medical experts continue to emphasize that the positive benefits of the COVID-19 vaccines almost always outweigh any risks, with immunization especially recommended for people living with dementia.
I am a member of a racialized
community and am hesitant to get
the COVID-19 vaccine. How do I
know it’s safe for me?
It is understandable for racialized individuals, particularly Black and Indigenous Canadians, to be hesitant to get the vaccine due to systemic racism and historical mistrust of the Canadian health care system. However, Canadians of colour have been disproportionally affected by COVID-19 due to social, economic and health disparities, and getting the vaccine can provide them with the greatest level of protection from this virus.
Furthermore, Pfizer-BioNTech and Moderna have said the efficacy of their vaccines has been consistent across age, race and ethnicity, and gender demographics.
Does getting the vaccine mean
that I will get COVID-19?
The COVID-19 vaccines that are currently available to Canadians cannot and will not give you COVID-19. All vaccines basically carry a recipe card that your body then uses to make just the small piece of harmless spike protein that is similar to the protein that exists on the actual COVID-19 virus, which helps your body recognize and fight the virus.
The COVID-19 vaccine does not actually contain the COVID-19 virus, so you cannot get COVID-19 from the vaccine. The synthetic DNA and mRNA that is central to the COVID-19 vaccines also breaks down quickly once it enters your body.
The COVID-19 vaccine will not make a nasopharyngeal (nasal) PCR test or Rapid Antigen Test (RAT ) positive. If you test positive for COVID-19 on a RAT or PCR test, this means you have a COVID-19 infection, and it is unrelated to the vaccine.
I heard the materials in the
vaccine are harmful. Is that true?
As noted earlier in this pamphlet, both the Pfizer-BioNTech and Moderna vaccines contain mRNA, whereas the Novavax vaccine contains SARS-CoV-2 recombinant spike protein and the adjuvant Matrix-M. Other ingredients in the approved COVID-19 vaccines are normal vaccine ingredients, such as fats, salts and a small amount of sugar. The vaccines do not include live virus and will not interact with our DNA.
All of the approved COVID-19 vaccines do not include any gelatin or animal materials. The Pfizer-BioNTech, Moderna and Novavax vaccines were not developed using fetal cell lines. It is important to note that none of the vaccines contain any fetal tissues or cells. The currently approved vaccines do not contain common allergens such as latex and food proteins (e.g., eggs, gluten, nut products or byproducts). The vaccines do not contain any material such as metals, implants, microchips or tracking devices.
I heard that individuals may get
myocarditis/pericarditis after
getting the COVID-19 vaccine. Is
that true?
There is a very rare risk of individuals getting myocarditis (inflammation or swelling/redness of the heart muscle) and/or pericarditis (inflammation or swelling/redness of the lining outside the heart) through the available COVID-19 vaccines. The symptoms for most people resolve quickly through standard treatment and rest. Also, it has been found for both Pfizer-BioNTech and Moderna vaccines that these cases occurred particularly in adolescents and young adults. It is important to consider that myocarditis can even be result of COVID-19 infections.
Individuals who experienced myocarditis or pericarditis from a previous COVID-19 vaccine, may still receive another dose of the vaccine after first discussing this further with their health care providers.
How many older adults have been
vaccinated in Canada?
As of summer 2023, 97 per cent of Canadians aged 60 years and older have received at least one dose of the COVID-19 vaccine, with 96 per cent having completed a primary series. However, only 15 per cent of older adults in Canada (not including Quebec) received a vaccine dose in the last six months. This indicates most older Canadians are potentially eligible to receive the updated XBB.1.5- containing COVID-19 vaccines. This vaccine is expected to provide a better immune response against the current circulating COVID-19 strains, and also improve protection against infection, symptomatic and severe disease that might have been reduced since a person’s last vaccination or infection.
How can I get a record of my
COVID-19 vaccination history?
There are different ways you can get a record of your COVID-19 vaccination history. The online method includes the following instructions:
- Click on the following website link: https://www.canada.ca/en/publichealth/services/diseases/coronavirusdisease-covid-19/vaccines/vaccineproof.html.
- Click on your province/territory box.
- Click on the green box to visit the jurisdictional website.
- Provide the needed information to obtain your COVID-19 proof of vaccination.
The telephone method includes the following instructions:
- Contact your primary health care provider as they should have a record of your COVID-19 vaccinations.
- If you are not able to confirm your vaccination history, ask your health care provider for other ways to try to determine this.
Will we have to continue getting
a vaccine dose every three to six
months?
Currently, there is uncertainty surrounding the annual pattern of COVID-19 infection, duration of immune protection from vaccines and/ or infections, and the impact of future vaccines. As more research and data is released on these topics, NACI will provide Canadians with further recommendations on the interval between vaccine doses.
However, individuals should not wait on these decisions and act now on receiving vaccines, especially when the fall season marks a time of heightened respiratory virus transmission and infections.
Are there other vaccines that I
should be getting?
There are five other vaccines approved and/or recommended for older adults in Canada, including:
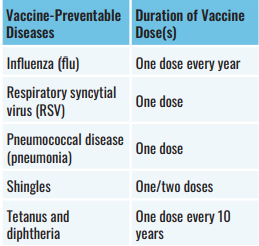
Most recommended vaccines can be given at the same time. Speak with your health care provider regarding staying up to date with your recommended vaccines.
To learn more about these vaccine-preventable diseases, national vaccine recommendations, costs and availability, please click on the following pamphlet link:
Modified: 2024-02
We would like to thank the following for their support of our Education Resources to Support Healthy Ageing Initiative

